X-ray 3D CT Reveals Magnetic Control of Catalyst Structure for Tunable Methane Oxidation
In a significant breakthrough for catalytic science, a research team led by Professor Yuen Wu at the University of Science and Technology of China has developed an innovative iron-embedded liquid metal catalyst (Fe–LMS) system that enables precise, reversible control over methane oxidation product selectivity using an external magnetic field. This approach provides a novel solution to the long-standing challenge of the "adsorption energy scaling relationship" in catalysis.
The study demonstrates that under ambient conditions, the Fe–LMS catalyst achieves remarkably high production rates and selectivities. By finely tuning the magnetic field intensity between 0 and 500 G, researchers can precisely control the product distribution. The system exhibits excellent reversibility, maintaining high selectivity over 11 switching cycles. This discovery not only breaks through the limitations of traditional "adsorption energy scaling relationships" but also offers a low-energy, operationally simple method for industrial catalysis, with significant potential to reduce the energy consumption and cost of producing high-value chemicals from methane oxidation.
The Critical Role of X-ray 3D CT
A key element in understanding and validating this magnetic control mechanism was the use of advanced synchrotron-based characterization. X-ray 3D computed tomography (CT), conducted at the BL07W beamline of the Hefei National Synchrotron Radiation Laboratory, played a crucial role in directly visualizing the catalyst’s structural response to the magnetic field.
The non-destructive, high-spatial-resolution imaging capabilities of X-ray 3D CT allowed researchers to observe the dynamic spatial distribution of iron atoms within the liquid metal droplets without damaging the samples. The in situ 2D CT slices and 3D reconstructions provided direct and compelling evidence: under zero-field conditions, iron atoms were uniformly and symmetrically distributed throughout the spherical liquid metal droplet. However, upon application of a 500-G magnetic field, the iron atoms distinctly aggregated toward one side of the droplet.
This direct visualization of iron atom migration and aggregation provided conclusive experimental support for the core hypothesis—that the magnetic field modulates the reaction pathway by controlling the aggregation state of iron atoms. The dispersed iron atoms (single atoms) favor the C1 pathway to CH₃OOH, while the magnetically induced iron clusters promote C–C coupling and the C2 pathway to CH₃COOH.
The application of X-ray 3D CT in this study not only validated the proposed mechanism of magnetic-field-driven catalyst reconfiguration but also established a powerful methodological framework for developing and understanding novel catalytic systems controlled by external fields.
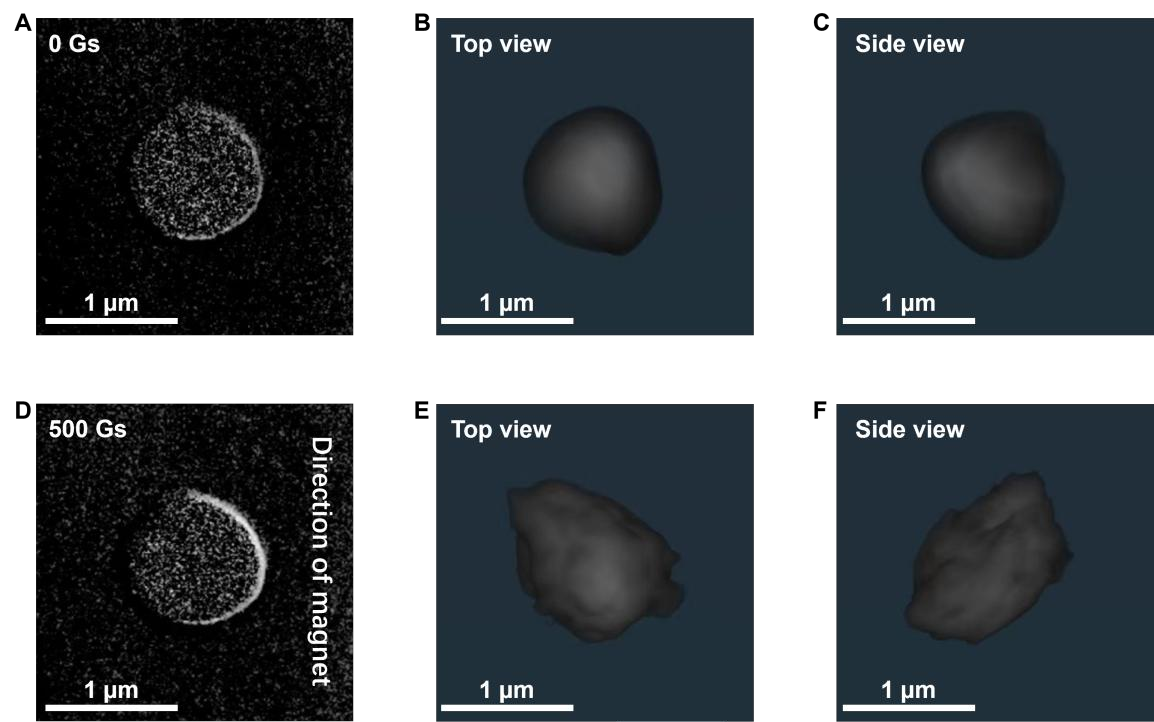
Figure 1 A–C. 2D and 3D CT reconstruction images of Fe–LMS under 0 G. D–F. 2D and 3D CT reconstruction images of Fe–LMS under 500 Gs. These images provide direct evidence of the magnetic field’s influence on iron atom aggregation: under 0 Gs, X-ray 3D CT reveals that iron atoms are uniformly and symmetrically distributed within the sphere (Fig. 1A); under 500 Gs, X-ray 3D CT shows that iron atoms distinctly aggregate toward one side (Fig. 1D).
This work is published in Nature Nanotechnology under the title “Magnetically tunable selectivity in methane oxidation enabled by Fe-embedded liquid metal catalysts.”
Full article: https://doi.org/10.1038/s41565-025-02029-5
Back

