New Progress in Electrocatalytic Nitrate-to-Ammonia Synthesis by Users of Hefei Light Source
Ammonia serves as an important basic chemical raw material in the national economy and is also a promising zero-carbon energy carrier in the future renewable energy system. Its green and sustainable preparation technology is crucial for achieving the “Dual Carbon Goals”. Nevertheless, the conventional Haber–Bosch process relies on high temperature, high pressure, and fossil fuel-derived hydrogen, leading to excessive energy consumption and carbon emissions that fail to meet future development requirements. The electrochemical approach, driven by renewable energy to split water for hydrogen supply, enables ammonia synthesis under ambient temperature and pressure conditions. In particular, nitrate (NO₃⁻) has become a significant technical route for both environmental remediation and resource utilization due to its lower activation energy barrier, good water solubility, and widespread presence in industrial wastewater. However, the nitrate reduction reaction (NO₃⁻RR) involves a complex multi-electron transfer process, and multiple nitrogen-containing intermediates (e.g., NO₃⁻, NO₂⁻, NO, NH₂OH, etc.) exhibit distinct adsorption properties and hydrogenation demands, making it difficult for conventional catalysts to possess both efficient deoxygenation capacity and precise hydrogenation capability. In addition, the hydrogen evolution reaction (HER), as a competitive reaction, further impairs the efficiency of NO₃⁻RR. How to simultaneously achieve “efficient NOₓ adsorption/activation” and “sustained *H supply” in a single catalyst system has long been a core scientific challenge in this field.
Prof. Wei Chen’s research group from the University of Science and Technology of China (USTC), relying on the in-situ micro-electrochemical infrared spectroscopy developed at the BL01B of Hefei Light Source, has made new progress in the field of electrocatalytic nitrate-to-ammonia synthesis. In this study, an integrated cascade catalytic system consisting of Fe single atoms and Ru nanoparticles with strong interfacial coupling and functional partitioning was constructed, successfully breaking the inherent limitation of mismatched intermediate conversion and hydrogen species supply during nitrate electroreduction, and realizing high-efficiency and high-selectivity ammonia production at low potentials. This research not only establishes a complete mechanistic evidence chain through operando spectroscopy and theoretical calculations but also provides a universally applicable strategy for catalyst design in multi-step and multi-electron reaction systems.
The research team utilized in-situ micro-electrochemical infrared spectroscopy to real-time monitor the dynamic evolution of intermediates on the catalyst surface during nitrate reduction. The spectroscopic results reveal that characteristic signals of NO₂⁻, −NH, Ru–H, and other species in different catalytic systems vary significantly with applied potential, directly reflecting the differences in reaction pathways and kinetics.
It is found that the introduction of Ru nanoparticles significantly weakens the signal of the NO₂⁻ intermediate and remarkably enhances the peaks related to −NH, indicating that the hydrogenation of nitrate-derived intermediates is effectively promoted. Meanwhile, the emergence of the Ru–H signal unveils the pivotal role of Ru sites in the generation and transfer of active hydrogen. This work elucidates the mechanism of synergistic regulation of active hydrogen behavior by Ru–Fe dual active sites based on in-situ infrared spectroscopic evidence, offering new experimental support for the design of high-performance nitrate reduction catalysts.
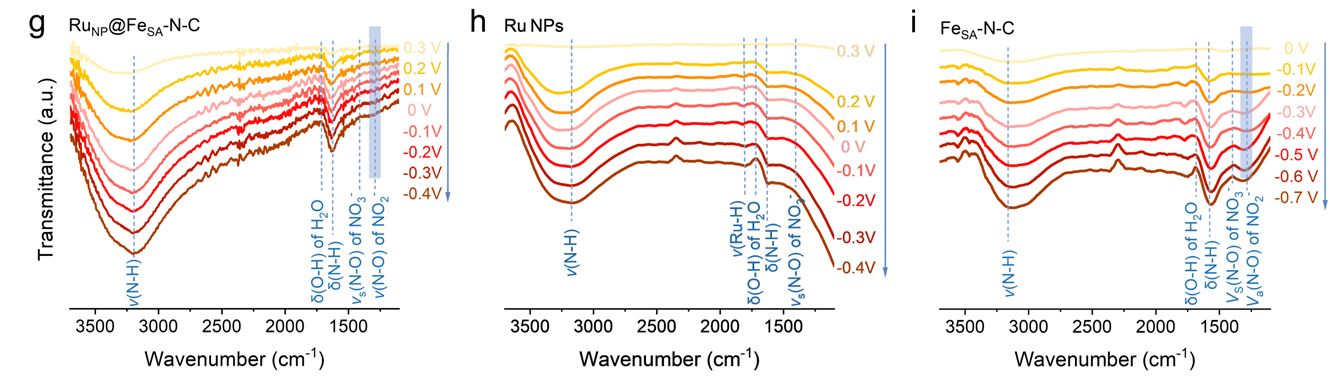
Figure 1. Operando FTIR spectra of (g) RuNP@FeSA-N-C, (h) Ru NPs, and (i) FeSA-N-C at different applied potentials during the NO₃⁻ reduction reaction in argon-saturated 1 M KOH + 0.1 M KNO₃ electrolyte.
The related research results were published in the renowned international academic journal Journal of the American Chemical Society (J. Am. Chem. Soc.) under the title “Integrated Cascade Catalysts for Electrochemical Nitrate Reduction to Ammonia”.
Back

